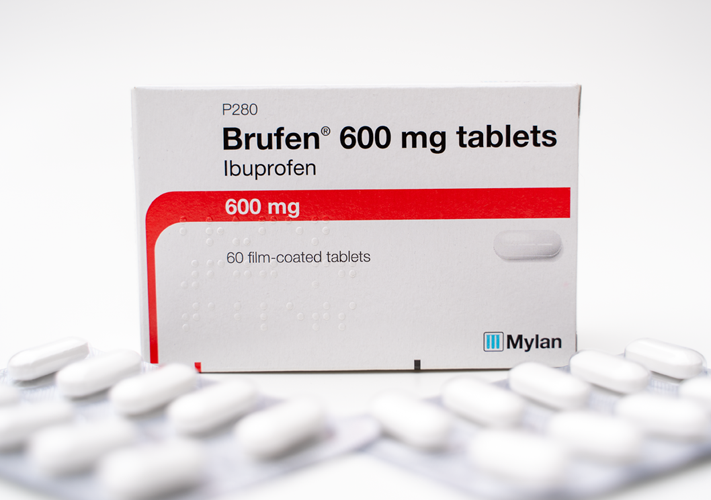
Testing ibuprofen solubility and dissolution in biorelevant media
Drug: ibuprofen tablets
Therapeutic category: Non steroidal anti-inflammatory
Release: immediate
Type: neutral
Food effect: none, only delayed Tmax
BCS classification: class 2A
In vitro biorelevant dissolution tests were conducted on ibuprofen tablets (Brufen forte®) to identify suitable biorelevant media to support the formulation development of a generic version of the drug.
Some of the biorelevant media were found to be unsuitable because they were insufficiently discriminative for the selection of the formulation. However, the results clearly identified other appropriate media and a method to identify bioequivalent formulations of this relatively high dose water-insoluble acidic compound. The reason for a delayed Tmax in the fed state also became obvious when the dissolution results were compared. This is important when considering the patient because although the frequently recommended co-administration of this drug with food can reduce gastric irritancy; a meal may delay onset of activity.
If you would like to discuss how these type of biorelevant tests will help you in the development and selection of your bioequivalent test product, please feel free to contact our highly knowledgeable Technical Support team here.



















 Home
Home















