HPLC linearity standards are prepared at a series of known concentrations (i.e. from 0% to 120%) to confirm that the detector response is directly proportional to drug concentration across the expected sample concentration range after dilution. This ensures that unknown dissolution samples after can be accurately quantified from the calibration curve.
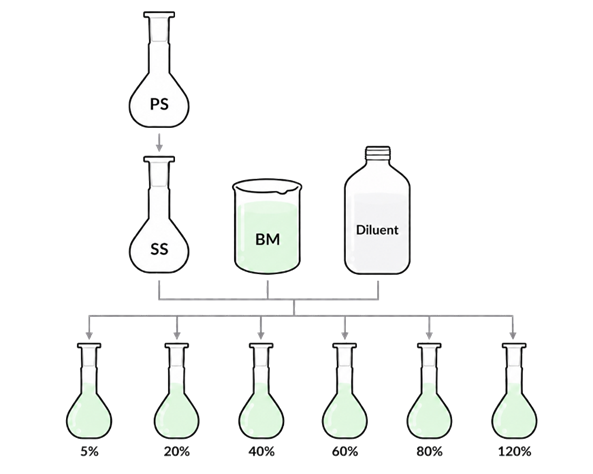
During linearity standard preparation, it is important to incorporate biorelevant media and use diluent (at the appropriate dilution ratio you have selected, typically dilution ratio is between 1:1 to 1:10) to evaluate any interference between the analyte peak and background peaks. The following parameters should be checked:
· R2 value
· Limit of Detection and Limit of Quantification
· Stability of sample after 24hours
If chromatographic performance is not satisfactory:
· Consider modifying the injection volume
· Consider modifying the dilution ratio
· Check UV lamp response and signal to noise ratio
· Check calibration of autosampler or manual injector



















 Home
Home















