What is drug supersaturation?
Supersaturation is a concentration of drug that is above the equilibrium value of its solubility in the selected test medium. The dissolved supersaturated molecules are in a high energy state compared to the lower energy state drug of a drug dissolved at equilibrium.
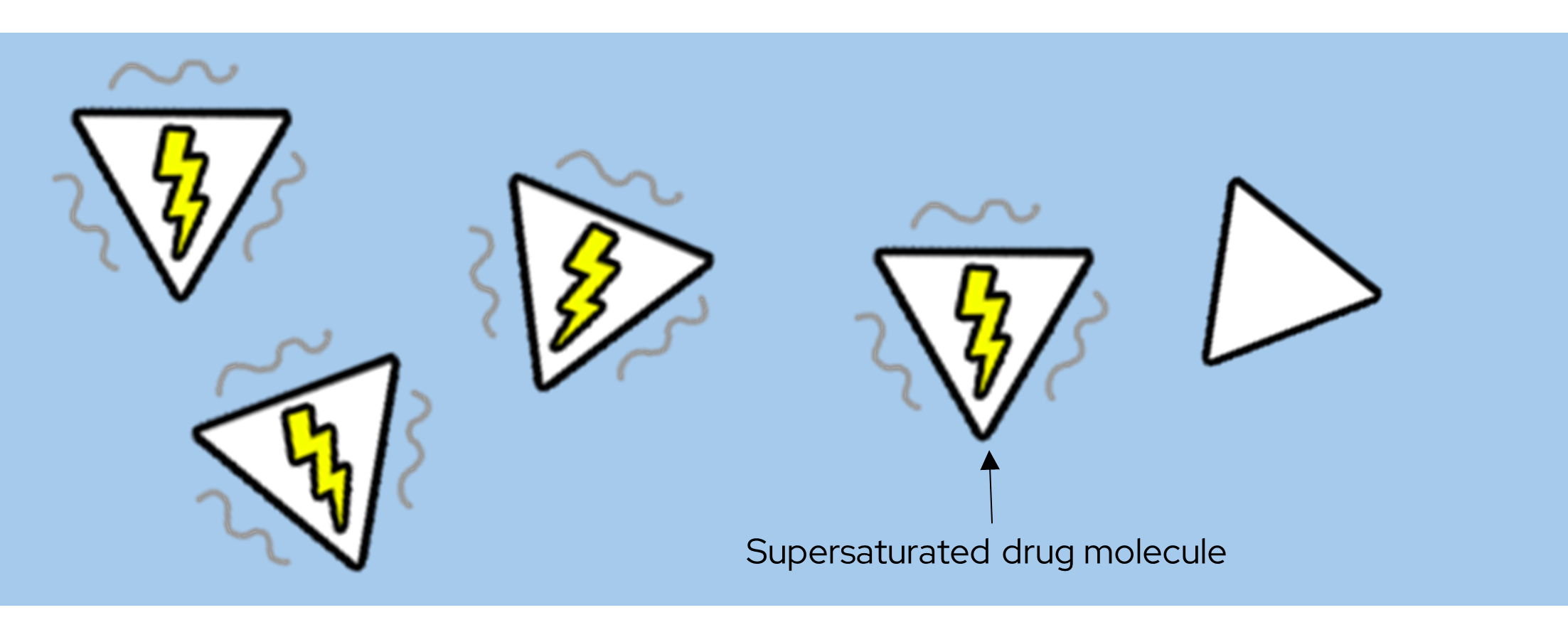
Metastable
Supersaturation is a metastable state of a drug in a test medium. Supersaturation can be achieved by, for example, manipulating the physical form or chemical form of a drug. Manipulation of a drugs physical form can be achieved via formulation such as creating a solid dispersion. The chemical form of a drug can be modified by use of a co-crystal former or a counter ion.
Improve drug absorption
The ability and ease of a drug to supersaturate (or not) in biorelevant media can provide a developer an indication how readily this property can be exploited to deliver a drug. Sufficient supersaturation of a poorly water-soluble drug in in vitro biorelevant media can indicate that its in vivo intestinal absorption can significantly improved if the drug were to be administered orally.
Test a formulated drug
When one wants to understand the role of excipients or formulation approach on saturation, testing the dissolution of the formulated drug in biorelevant media can be especially useful. The results can reveal the supersaturation or crystallisation behaviour within intestinal fluids from which absorption occurs enabling a developer to monitor the magnitude of the supersaturation and how long the drug remains in a supersaturated state. The results from dissolution tests are much more useful a result from a single solubility time point.



















 Home
Home















