Dissolution of a neutral drug in fasted state biorelevant media
-
Carry out entry level dissolution testing in FaSSGF and FaSSIF which simulate human fasted state gastric and intestinal fluids
-
Gain crucial insight into how gastrointestinal fluids influence drug release and solubilization
-
Support oral drug developability assessment and formulation development
Background
For an orally administered drug to be absorbed, it must first be released from its formulation and dissolve in the fluids present in the gastrointestinal tract. The way a drug behaves in fasted state gastrointestinal fluids can strongly influence how much drug and how quickly the drug becomes available for absorption after administration with a glass of water. To investigate this process, simple in vitro dissolution tests can be performed using biorelevant media. These tests use conventional HPLC analysis and USP dissolution apparatus with vessels containing media that simulate the fluids found in the stomach and upper intestinal tract under fasted state conditions. There is no need for specialist equipment.
MATERIALS




FaSSIF is made with 3F Powder and FaSSIF Buffer Concentrate

FaSSGF is made with 3F Powder and FaSSGF Buffer Concentrate

Available in boxes of 72 pieces
RECOMMENDED BUYS
1 x FFF, 1 x FASGBUF, 1 x FASBUF and 2 x GMF13 for 6 dissolution vessels of FaSSGF (900mL) and 6 dissolution vessels of FaSSIF (900mL).
METHOD

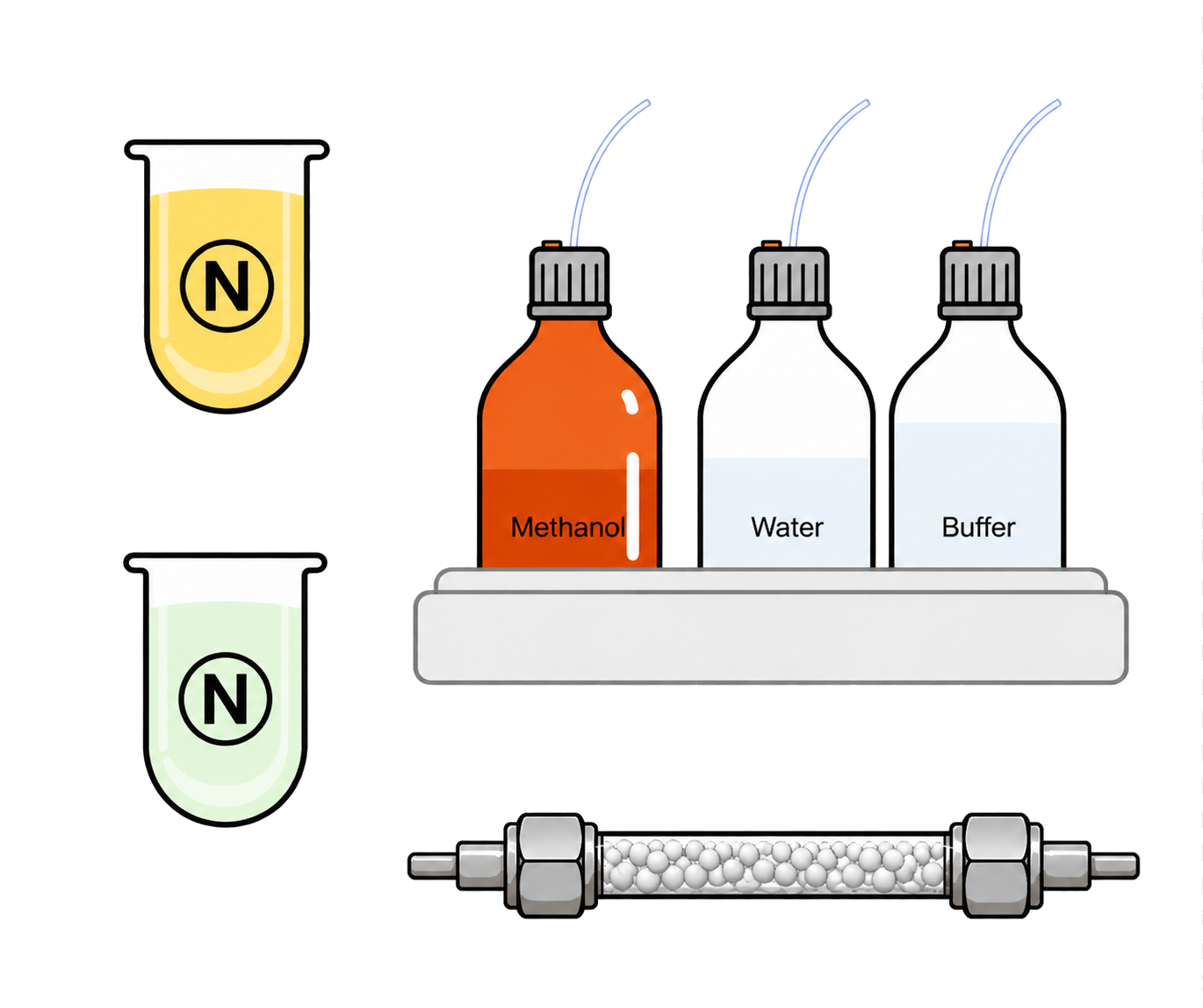
- Use HPLC for drug analysis
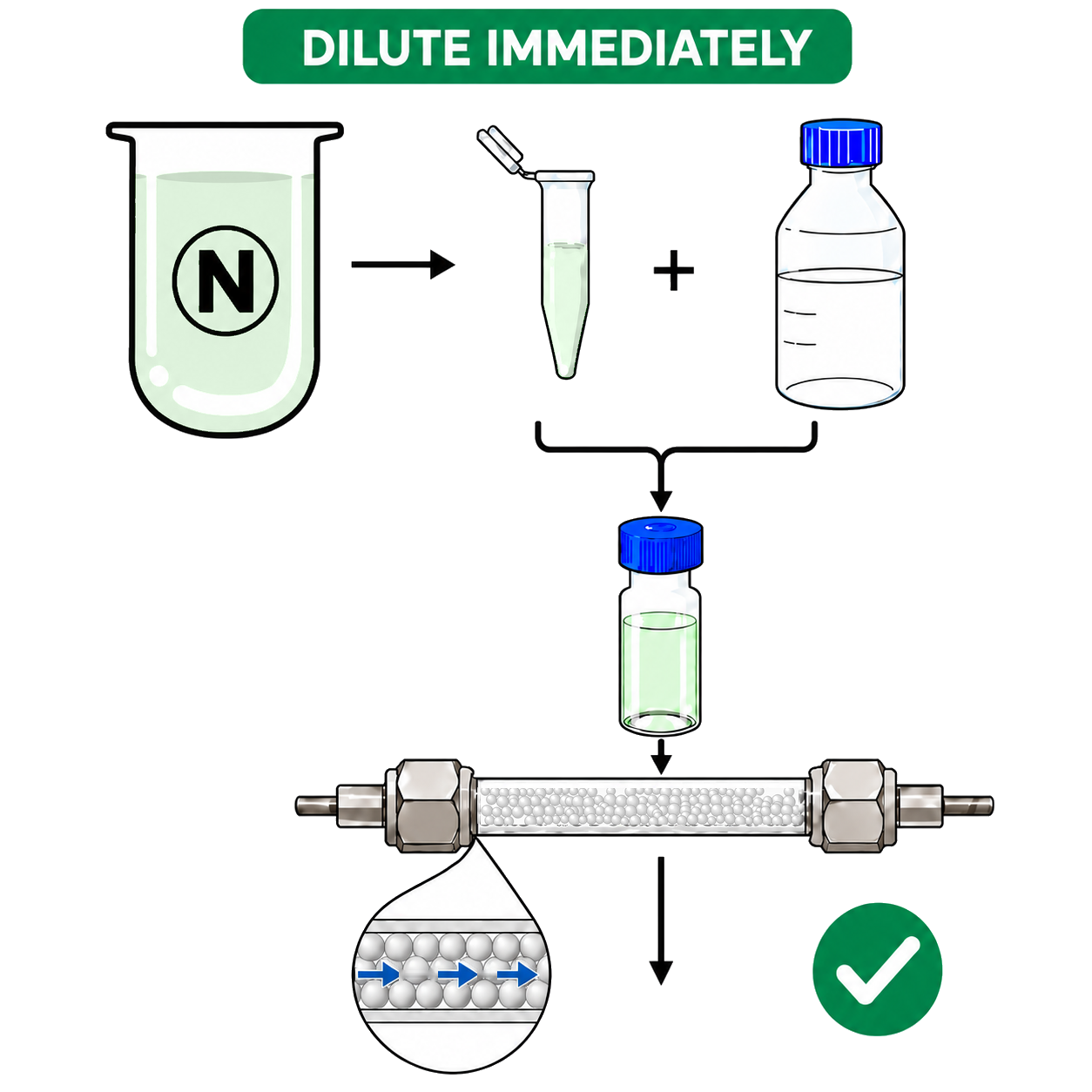
- We recommended diluting samples immediately
- Use a methanol-based diluent for sample dilution
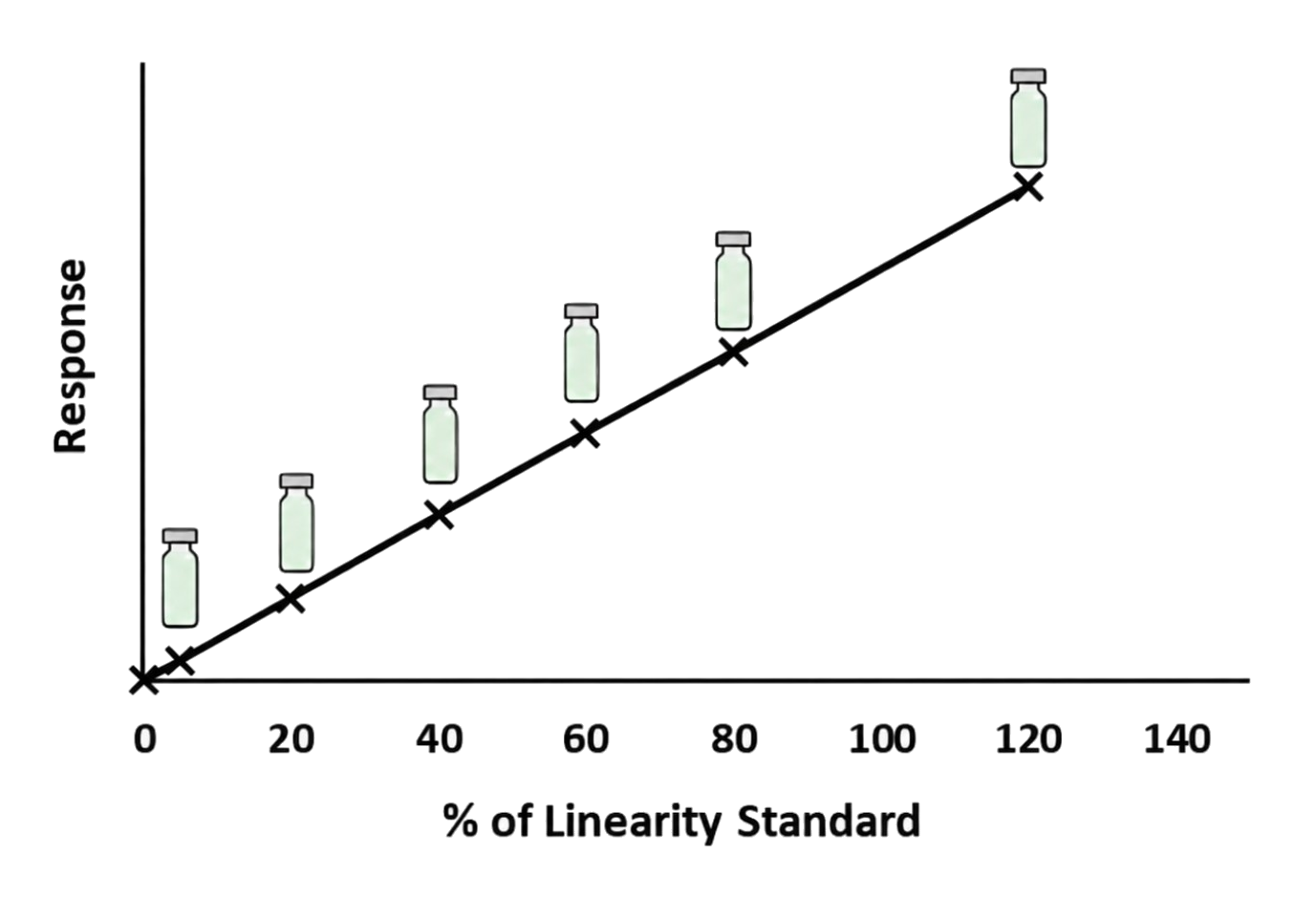
- Establish linearity and determine LOD and LOQ
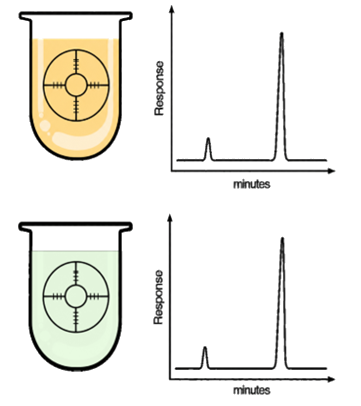
- Check method specificity and assess precision
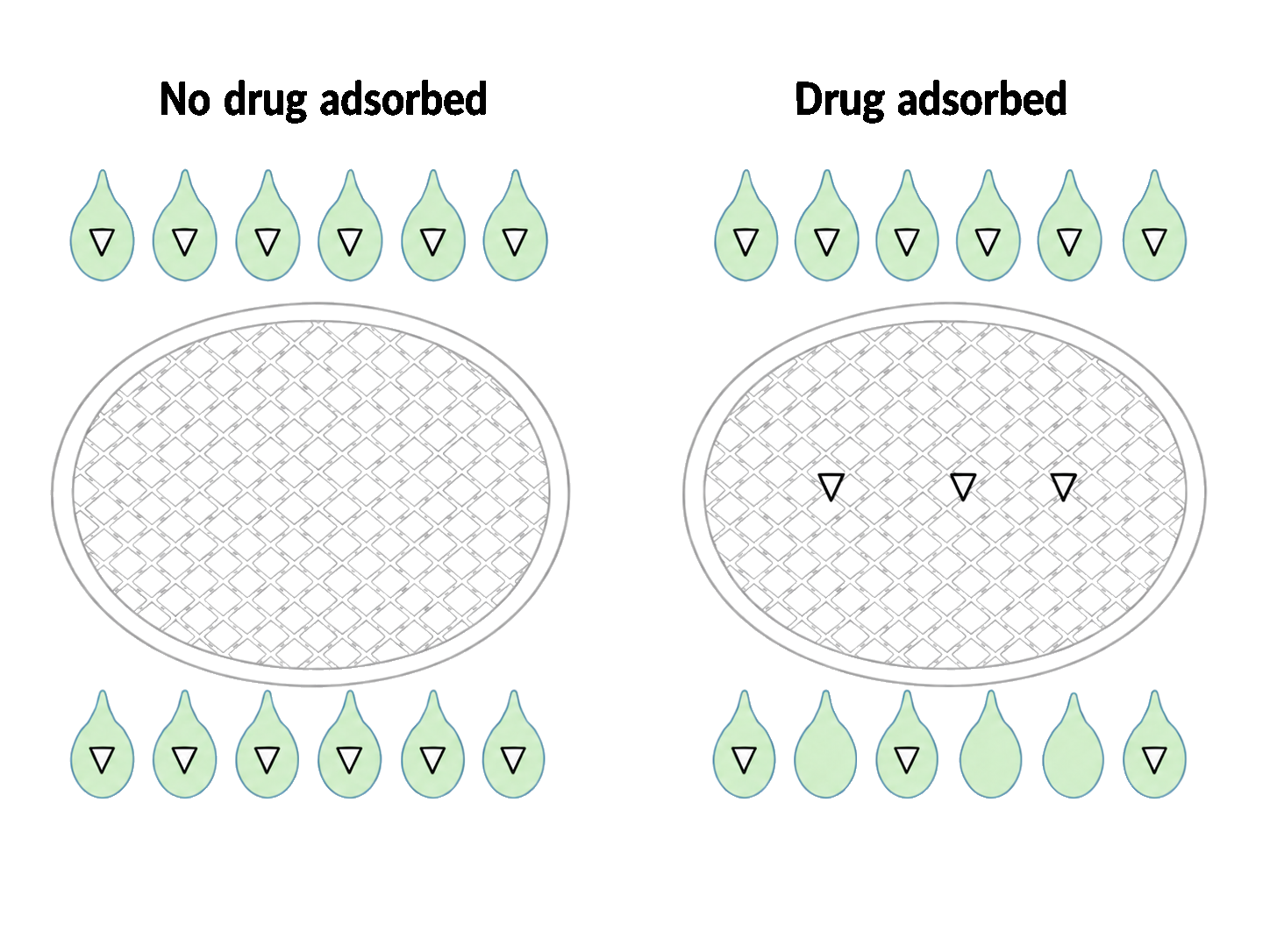
- Biorelevant Dissolutions Filters (GMF13) are recommended with a sampling volume of 5mL
- Filter adsorption establishes pre-saturation volume required for sampling
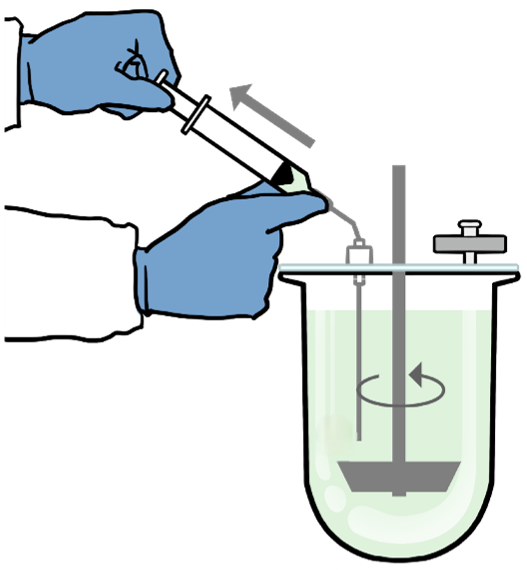
- Perform dissolution testing using the selected biorelevant medium
- USP Dissolution Apparatus 2 is recommended, typically operated at a paddle speed of 75 rpm or higher, to ensure homogeneous mixing
- 900mL is typically recommended as the starting test volume
- Compared to QC testing, longer sampling time points are generally recommended
- Use fresh filters to increase accuracy of results by stopping dissolution and preventing carry over from previous sampling



















 Home
Home