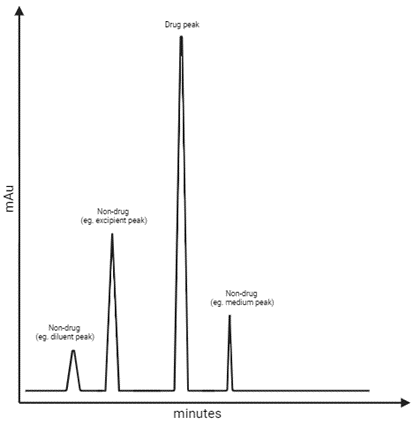
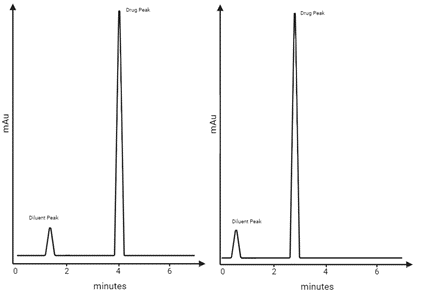
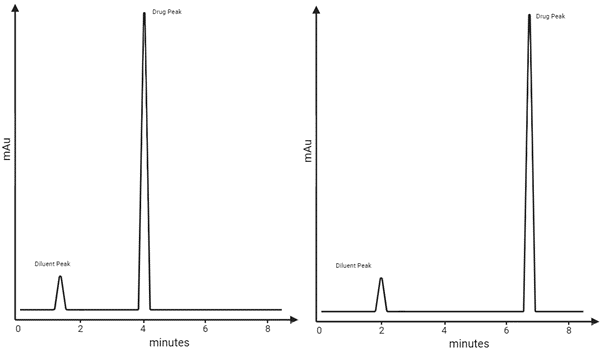
Learning Centre
ALL
NEW!
Background
Biorelevant media
Analysis
Filters
Dissolution tips
Guides and Calcs
Dissolution
















 Home
Home